Successful Cancer Screening Depends Upon Trust
Why Routine Measures for Early Cancer Detection are Failing to Save Lives
Rethinking Cancer Screening
I. Introduction
Recently, a seventy-two-year-old patient in our practice posted a negative review online. He had come in for an annual physical expecting to be screened for prostate cancer but his primary care physician (PCP), who is board-certified in internal medicine with more than forty years of clinical practice experience and up-to-date on the latest evidence-based recommendations for cancer screening, counseled him against it, explaining that the risk/benefit ratio for that test, called the prostate-specific antigen (PSA) test, is poor. We now know that for men in their seventies, prostate cancer screening is correlated to both shorter lifespans and healthspans (years of life spent in good health) compared to those who do not get screened.
If that doesn’t make intuitive sense to you, you’re not alone. How can it be true that catching cancer earlier rather than later (the point of screening tests) is associated with worse, not better outcomes? The answer is nuanced and will be discussed shortly but first, I wanted to focus for a moment on the patient in question who arrived at our office wearing the armor of skepticism rooted in a widely-held narrative about today’s healthcare system that frames physicians as unscrupulous entrepreneurs motivated mainly by profit. Through that lens, the discussion he had with his PCP was not experienced as an exceptionally good interaction with a caring, informed doctor about the latest and best understanding of the value of PSA testing, but was instead viewed as part and parcel of a culture of medical negligence and/or incompetence driven by corrupt incentives—a story that he felt compelled to channel into a scathing online review that included the following: “I guess he doesn’t make enough money on the test.”
Fortunately, this is not an everyday occurrence for us. Our practice has been in the same neighborhood for close to four decades, during which time we have worked very hard to build deep relationships of trust within our community. Unfortunately, however, such things are no longer exceedingly rare either. To understand why, we must begin by stepping back far enough from a small story of one disgruntled patient to see (at least the outlines of) a much larger one informing American society today—one that has helped usher in an era of pervasive mistrust in our basic societal institutions, including science and medicine.
This is a complicated narrative deserving of a separate thought piece that explores in detail, in an unbiased way, the unintended missteps of our premier public health agencies, especially during the pandemic; the stubborn persistence of thought leaders in mainstream medicine to focus on relieving or managing diseases rather than preventing them; the corporatization of hospital systems and medical groups using a strategic model that prioritizes profit over the well-being of patients and healthcare workers (especially, ironically, PCPs); the infiltration of science by the corrupting forces of money and politics; the ready availability of unvetted medical information online that has been harnessed in recent years to fuel the so-called medical freedom movement in America; and perhaps most importantly of all, the explosive proliferation (again, especially during the pandemic) of corrupt actors within the burgeoning industry of false conspiracy theories peddled on social media and so-called ‘alternative news’ platforms in print and on television, directed not just against the government but against technical experts of all kinds, including those in science and medicine, which is steadily gaining market share in the competition for our attention through a business model of driving outrage.
It seems fair to say that today, a growing share of medical providers feel like they are often working from a deficit to earn the respect and trust of patients who have come to view the world through a cynical belief construct that has its roots, at least in part, in conspiratorial misinformation and disinformation.
To successfully bridge that gap, today’s PCPs must be up to date on both the most current medical science and the latest pseudo-scientific fads and trends; they must possess special skills and personal qualities that enable them to share nuanced understandings with patients in a way that is persuasive, non-judgmental, and relatable; they must be able to cut across barriers of politics and pre-held suspicions in a way that does not trigger defensiveness. And all too often, this must take place within the constraints of a fifteen or twenty-minute office visit.
That’s a lot to ask of healthcare providers who are under more pressure than ever to practice medicine in a way that satisfies today’s healthcare overlords—the hospital, medical group, and insurance company administrators in whose hands most of the power in mainstream medicine has been concentrated. This too is worthy of a separate thought piece and I don’t want to shoot too many arrows all at once. Suffice it to say that things have changed since I began my practice thirty-six years ago.
Before the advent of managed care, respect for and trust in doctors was high, and a knowledgeable, experienced internal medicine physician who always operated out of the best of intentions could feel confident sharing information with and making recommendations to his or her patients with the expectation that they would feel well cared for and appreciative. Ironically, the added work facing many PCPs today of shepherding facts, advice, and nuanced understandings of best practices for things like cancer screening across a well-defended barrier of suspicion rooted in conspiratorial disinformation is beyond their abilities precisely because they are not clever entrepreneurs skilled at the art of deception, but are instead, for the most part, caring, sensitive, overworked, often anxious, chronically sleep-deprived, slightly-nerdy moms, dads, and grandparents who are, by their very nature, focused on taking care of others, not on fleecing their patients.
With that in mind, let’s now turn our attention to the subject of this Health and Science Briefing: cancer screening.
II. PSA-Testing, for Example…
Cancer screening is an attempt to catch malignancies in their early stages when they are more treatable and, hopefully, curable. This makes perfect sense and I am certainly not making an argument that cancer screening is a bad practice. But, insomuch as the deepest objective of early cancer detection is to extend the lives and well-being of patients, screening involves quite a bit more than simply running tests designed to detect cancer during annual physicals or according to some other broadly predetermined timeline.
The first thing we need to really understand is that cancer itself is complicated and nuanced, and so, therefore, is when and how to look for it and what to do when we find it. Critically, not all cancers shorten lives or even make people sick. Some forms of cancer grow so slowly and spread so infrequently that they rarely cause harm, especially when discovered in later life.
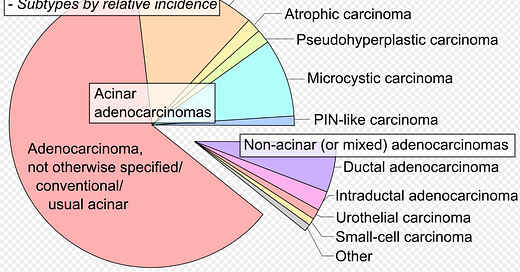
For example, the most common form of prostate cancer called acinar adenocarcinomas (AA), tends to be very slow-growing and rarely ever spreads outside of the prostate. AA is most commonly diagnosed among men in their late sixties and carries a >99% survival rate over five years and a 95% survival rate over fifteen years. A new diagnosis of acinar adenocarcinoma in a seventy-two-year-old man has about a 5% chance of harming his health by his eighty-seventh birthday [If your mind just jumped from that statistical unpacking to the thought that a five-percent chance of harm is not zero and that, therefore, you would still very much want to know if you had acinar adenocarcinoma of the prostate, hold that thought].
PSA goes up significantly in patients with AA. However, other far less common but far more aggressive forms of prostate cancer often fly under the radar of that screening test (at least until they have progressed quite far). In other words, the PSA screening test is far more likely to detect a slow-growing and relatively non-dangerous acinar adenocarcinoma than a much more aggressive, non-acinar adenocarcinoma.
That understanding has some important ramifications. Abnormally high PSA levels may be an early indication of cancer but when it is, more than 95% of the time it’s the kind that rarely causes illness or becomes life-threatening and is best kept under surveillance and not treated. But how many men in the modern era of patient-directed care, are willing to live with knowing that they have cancer and just watch it over time? What are the psychological effects of cancer surveillance? And what percent of men, against the recommendations of their doctors, nevertheless push for aggressive, high-risk treatments in the hope of achieving the peace of mind of knowing that their prostate cancer has been eradicated (keep reading)?
In short, AA caught on a PSA test can lead some men down the road toward harmful overtreatment. At the same time, normal PSA levels can create a false sense of security since far more aggressive non-acinar adenocarcinomas of the prostate often fail to cause abnormally elevated PSA levels. A positive PSA test is not proof that a patient’s health is at risk and a negative PSA test is not proof that a patient does not have an aggressive form of prostate cancer. I hope that you’re already starting to get a sense that a comprehensive understanding of the value of this particular cancer screening test is complicated and nuanced. Hold on, things are about to get more complicated…
Elevated PSA Can Have Many Causes
Adding to the complexity of prostate cancer screening is the fact that AA is not the only thing that drives PSA out of the normal range. Inflammation of the prostate (prostatitis) can also do it. Sometimes prostatitis is triggered by an infection but in many cases, the cause is unknown. In addition, mechanical pressure on the prostate such as occurs during bicycle riding, having a prostate exam (part of a routine annual physical for men over the age of fifty), or ejaculation can elevate PSA. So can older age, an anatomically larger than average-sized prostate, urinary tract infections, and a condition called benign prostatic hypertrophy (BPH) which is common among (even healthy) men over the age of fifty. All told, about three-fourths of men over fifty with high PSA levels (above 4 ng/mL) don’t have prostate cancer.
Let’s pause here to sum things up so far:
Non-dangerous cancers that are best to watch, not treat, are likely to be caught by PSA testing and this can lead to aggressive and unnecessary treatments in the era of patient-directed care.
Dangerous cancers often go undetected by PSA testing in their early stages and this can lead to a false sense of security.
The many non-cancer-related drivers of elevated PSA levels collectively represent an additional confounding factor for doctors trying to make sense of this cancer screening test.
Back To Our Disgruntled Patient
As a thought experiment, let’s walk through some likely scenarios for what might have happened had the patient in question been given the PSA test that he came in expecting at the time of his annual physical.
About eleven percent of men over the age of seventy will have abnormally high PSA levels when screened, so, statistically, we would expect his test to have been normal. That would have eliminated subclinical prostatitis or a non-dangerous form of prostate cancer from his list of potential worries but it would not have eliminated the possibility of a more dangerous form of prostate cancer.
Had he been among the approximately one in ten men over seventy with PSA levels over 4.0 ng/mL, things might well have gone very differently. Men with PSA levels:
Below 4 have a 15% chance of prostate cancer.
Between 4 and 10 (the borderline range) have a 25% chance of prostate cancer.
Above 10 have a greater than 50% chance of having prostate cancer.
Had his PSA been abnormally high, the next step for most physicians would likely be to repeat the test or order a more sophisticated PSA assay called a prostate health index (PHI) which uses a mathematical formula combining subfractions of PSA (total PSA, free PSA, and proPSA) to calculate the risk of more worrisome forms of prostate cancer.
But neither standard PSA nor PHI are conclusive tests and a biopsy is often eventually ordered—even when the results do not indicate high-grade cancer (more about this later). Even biopsies, which involve snipping away tiny pieces of tissue for evaluation under a microscope, are not foolproof. Sometimes they snip the wrong areas and miss the cancer. Knowing this, and bearing in mind that high PSA can come from things other than prostate cancer, clinicians will sometimes prescribe a course (or perhaps multiple courses) of antibiotics followed by serial PSA testing, If PSA levels return to normal after antibiotic therapy, we can deduce that the cause of the elevated PSA was in infection.
However, given the panoply of factors that can drive elevated PSA levels, the likelihood that a course of antibiotics would have done the trick, especially in the absence of discomfort or other signs of infection, would have been low. Additionally, although antibiotic therapy is a relatively safe form of treatment, it is not entirely benign and carries side effects ranging from stomach aches to severe allergic reactions. Additionally, although they are our best weapon against bacterial pathogens, antibiotics also kill ‘friendly bacteria’ that live throughout our bodies, such as in the gut microbiome (GMB) upon which good health vitally depends. Dysregulation of the GMB by killing off friendly bacteria through repeated use of antibiotics can affect heart health, brain health, blood sugar regulation, immune health, and a host of other critically important physiological processes.
Starting down a diagnostic and/or therapeutic road initiated by elevated PSA can be stressful. And it can come with some health costs. And those costs tend to mount the further down the road one goes, as we will see shortly.
Let’s imagine that, as expected, antibiotic therapy failed to return his PSA levels to normal. If the repeat PSA or a PHI test were abnormal, the next step would likely have been to refer him to a urologist who would likely then perform or order a transrectal ultrasound study or (better) an MRI evaluation of the prostate.
Around ten percent of men between sixty-five and seventy-four have abnormal findings on an MRI even if they don’t have cancer. That share rises to eighteen percent among men seventy-five to eighty-four, and twenty-two percent among men eighty-five and up. Statistically speaking, we would expect nothing too suspicious to show up on imaging and the work-up for cancer would likely stop there. By this point, antibiotic therapy would have ruled out infection and a good history would have ruled out mechanical causes of prostatitis. After weeks or months of testing, treatments, and worry, he would be left with the unsatisfying ‘diagnosis’ of elevated PSA of unknown cause. Not the end of the world but it might leave someone like him wondering if perhaps he had been put through a money-generating mill of testing and treatment—especially if he typed a query into his internet search engine about high PSA and read that it isn’t even recommended for men his age.
For the sake of this mental exercise, however, let’s imagine that there had been some abnormal findings on his MRI. The next likely step in the context of elevated PSA that is non-responsive to antibiotic therapy and abnormal findings on MRI would be a biopsy of the prostate—a costly and very unpleasant procedure that almost always causes pain, discomfort, and difficulty with urination, at least for a while afterward, and carries the risks of causing local infection, bleeding, and in rare cases, more serious and even life-threatening outcomes such as sepsis.
If the biopsy results had come back negative (no cancer) our patient would have undergone the following medical journey:
One (or multiple) rounds of antibiotic therapy.
Repeat blood tests.
Referral to a specialist.
Imaging (MRI).
Biopsy.
Even if the side effects and costs accrued thus far had been acceptable to the patient in question, the weeks or months of worry associated with getting to that point are worth considering. In addition, despite all of that investigation, he would still not be able to be one hundred percent certain that he did not have cancer. Well, you might be thinking, what if the biopsy had come back positive for malignancy? Wouldn’t it all have been worth it to catch the cancer earlier rather than later? Not necessarily, keep reading…
Had the biopsy demonstrated malignancy, the likelihood that his cancer would be of the slow-growing, non-aggressive AA variety would have been over 90%. Since that kind of cancer rarely spreads and often does not cause significant illness in older men, the recommended next step would likely have been to keep good surveillance of the prostate through ongoing exams, blood tests, and imaging (surveillance). But the word ‘cancer’ causes some patients to react emotionally, not rationally, and in the new era of patient-directed medicine in which patients generally have the final say in medical decision-making, even if it goes against the recommendations of the physician, that can lead to unnecessary, aggressive, expensive, and dangerous interventions such as surgeries.
Since PSA testing began in the 1990s, an estimated six hundred thousand men have had surgery or radiation therapy to treat prostate cancers that would never have significantly harmed them. Approximately five hundred of those men died as a result of treatment while an estimated three hundred seventy-five thousand have experienced long-term erectile dysfunction, one hundred twelve thousand have experienced long-term urinary incontinence, and eighty-four thousand developed trouble controlling their bowels—all from treatments that experts now agree were unnecessary.
I hope, at this point, you are beginning to see why our patient in question was counseled against the PSA test and how it can be possible that cancer screening can drive bad outcomes sometimes. The U.S. Preventive Services Task Force (USPSTF) has found that screening for prostate cancer in men fifty-five to sixty-nine leads to high rates of overdiagnosis and overtreatment which, on balance, is causing more net harm than good. They give PSA testing a “C” rating for men in that age group (meaning no recommendation) and advise patients to talk to their doctors about whether or not to have the test based on their personal risk factors (such as genetic testing, past medical history, and family history). For men seventy and over, they assign PSA testing a “D” rating (D = don’t do it). Nevertheless, according to the CDC, in 2018, more than six million men over the age of seventy-five had a PSA test that year anyway. Some of them went on to have unnecessary treatments that shortened their healthspans and/or lifespans.
The data are speaking in a loud voice: elevated PSA among older men, in most cases, should be monitored, not treated. But in the US only about 60 percent of patients follow this surveillance protocol. One long-term study of 1,800 men with low or very low-risk prostate cancer found that within the first ten years of surveillance, almost half had ultimately elected to have aggressive treatments even though their risk of metastasis or death from prostate cancer was 0.1 percent over fifteen years.
II. Other Types of Cancer Screening
Since mammography began to be widely used to screen for breast cancer in the late 1970s, roughly 1.2 million women have had surgery for low-grade ductal carcinoma in situ (DCIS)—slow or non-growing cancer cells that rarely spread from the breast and experts widely agree are best to monitor, not treat. Approximately a thousand of them died and roughly a quarter of a million have experienced long-term post-mastectomy pain syndrome (intractable pain in the chest after removal of the breast) as a result of treatments that were arguably far more aggressive than what was needed.
Since the mid-1990s, when ultrasound screening for thyroid tumors began, surgeries for growths that never would have caused harm have killed roughly seven hundred patients. More than two hundred thousand have been left with damaged salivary glands causing pain and difficulty with eating, swallowing, and speaking. Thirty-four thousand needed surgery to repair damaged vocal cords. More than five thousand were left with some degree of permanent speech disability. And an estimated thirteen thousand have experienced permanent hypothyroidism with attendant muscle pain, fatigue, hair loss, dry skin, and depression.
III. The Big Picture: How Effective is Cancer Screening?
So just how beneficial is cancer screening? We can measure its effectiveness in terms of something called life years which quantifies the average number of additional years a person can expect to live as a result of a particular medical intervention such as a medicine, surgical procedure, vaccine, or, in this case, from following the recommended cancer screening guidelines. Before reading the next paragraph, take a moment and ask yourself the following question: How many additional life years are enjoyed by patients who follow the medical community’s standard of care protocols for cancer screening?
A new meta-analysis of the best (RCT) studies looking at mammography, colonoscopy, sigmoidoscopy, fecal occult blood testing, chest CT (for lung cancer in smokers and former smokers), and PSA testing, published recently in JAMA, shows that colonoscopy was by far the most effective cancer screening tool that we have. How many additional life years were added to the group who participated faithfully in colorectal cancer screening compared to those who never got screened? 0.4—a mere three months. No benefit in lifespan was seen for any of the other cancer screening tests. The American Academy of Family Physicians and the National Breast Cancer Coalition are starting to warn people that routine cancer screening may lead to harm.
In aggregate, patients who fall outside the USPSTF recommendations but get screened anyway—those who, according to the most current research, are more likely to be harmed than helped by cancer screening—cause an estimated $16 billion each year for private insurance and $4.6 billion for Medicare, for a total of $21.5 billion per annum. And that doesn’t take into consideration the personal costs paid by those patients in terms of healthspan and lifespan…
A New Blood Test for Colon Cancer
A new study published earlier this month in The New England Journal of Medicine showed that a new blood test to detect the DNA of cancer cells in the colon, called Shield, caught 87% of cancers while still at an early, curable stage. The false positive rate (the percent of test results that are falsely abnormal) was 10 percent. Using standard criteria, a test may be considered reliable if the rate of false positives is below 7.5% and the rate of false negatives is below 25%. Shield fails to meet those criteria. That said, a blood test is easier and far less expensive to perform than a colonoscopy and carries lower risk. In the hands of a physician trained in medical science, this new test has potential value when used on the right patients. However, because patients sometimes make emotional decisions, it also poses potential risks. Maximizing its value and minimizing its risk relies on three things: expert consensus recommendations based on science; well-informed clinicians; and patients having enough trust in their doctors’ recommendations to follow them without incurring debilitating levels of psycho-emotional stress.
First, Do No Harm
The idea that life-threatening cancers are easier to treat and more amenable to being cured if they are caught early rather than late makes so much intuitive sense that when it comes to cancer screening tests, it may be natural for patients to imagine there to be no downside to the practice of cancer screening. However, years of carefully collected health data have proven that overdiagnosis in the context of patient-directed healthcare is a driver of unnecessary and costly interventions that can lead to shortened healthspans and lifespans, negating the very reason for doing cancer screening in the first place.
The word cancer can derange rational decision-making for many of us. It can also be a source of ongoing stress and it has been well-demonstrated that chronic psycho-emotional stress can induce a state of chronic low-grade generalized inflammation (CGLI) which is harmful to our health. Months or years of worry about non-dangerous cancers like AA or DCIS can lead to major depression and drive other serious chronic psychiatric and non-psychiatric diseases. Such a comprehensive understanding must inform the education and decision-making processes that take place between doctors and their patients.
Let’s be clear: I am not making a blanket case against cancer screening. Instead, this Briefing puts forward an argument for a more selective and individually tailored approach that relies on and factors in the value of relationships of trust and respect between healthcare providers and patients. Algorithms that take into account a patient's symptoms, lifestyle, and health condition including comorbidities, age, past medical history, family history, and genetic testing will no doubt be dramatically improved by AI in the months and years to come. In the meantime, here is a quick reference guide for some common cancer screening tests:
USPSTF recommends that mammography every other year is beneficial for women between the ages of fifty and seventy-five.
They give screening for colorectal cancer for men and women between the ages of fifty and seventy-five an “A” rating (strongly recommended); screening for those between forty-five and forty-nine gets a “B” rating (okay but talk to your doctor first); and screening for those over seventy-five gets a "C" rating (more likely to cause harm than to help but talk to your doctor first).
They give PSA testing for prostate cancer in men between the ages of fifty-five and sixty-nine a “C” rating, and for those seventy and over a “D” rating (D = don’t do it).
Recommended Further Reading:
https://www.statnews.com/2022/02/24/more-more-more-approach-cancer-screening-harmful/


Hey Dr. Berkoff, I’m curious what you think about the GRAIL Galleri test. I am particularly curious about your perspective on their comparison of their false positive rate to that of serial mammography. They claim that if we assume mammography has about a 10% false positive rate, then a biannual screening program for a woman from ages 50 to 60 exposes her to a cumulative false positive rate of 50% through that decade. They compare this to their lower false positive rate, which is a sort of pooled analysis for all types of breast cancer, at least for what is published so far. What do you think of evaluating false positive risk in aggregate?
While I share your sentiment about the importance of trust in the physician-patient relationship, I’m also curious about your clinician perspective on “worried well” patients generally. Is it sometimes rational for a physician to offer additional screening simply because the patient is concerned, is likely to be well, and also likely to be placated by additional screening? Is it sometimes rational for the clinician to sort of treat the worry, particularly from a presumption of wellness?
Excellent article about the hazards of over testing for cancer.